21 February 2023
News
The network announces the publication of the DUPI-OEIL trial
Multicenter prospective observational study of dupilumab-induced ocular events in atopic dermatitis patients
Although ocular adverse events are frequent in AD patients treated with dupilumab, their characterization remains limited due to a lack of prospective studies with a systematic ophthalmological examination.
Our objective was to examine the incidence, characteristics and risk factors of dupilumab-induced ocular adverse events..
To do this, we designed a prospective, multicenter, and real-life study in adult AD patients treated with dupilumab.
At treatment initiation 27 out of 181 patients included had conjunctivitis. After 16 weeks of treatment, 25 out of 27 had improved their conjunctivitis and 2 remained stabl.
34 out of 181 patients had dupilumab-induced blepharoconjunctivitis (inflammation of the eyelid): either of spontaneous onset (n = 32) or worsening of underlying blepharoconjunctivitis (n = 2), figure below.
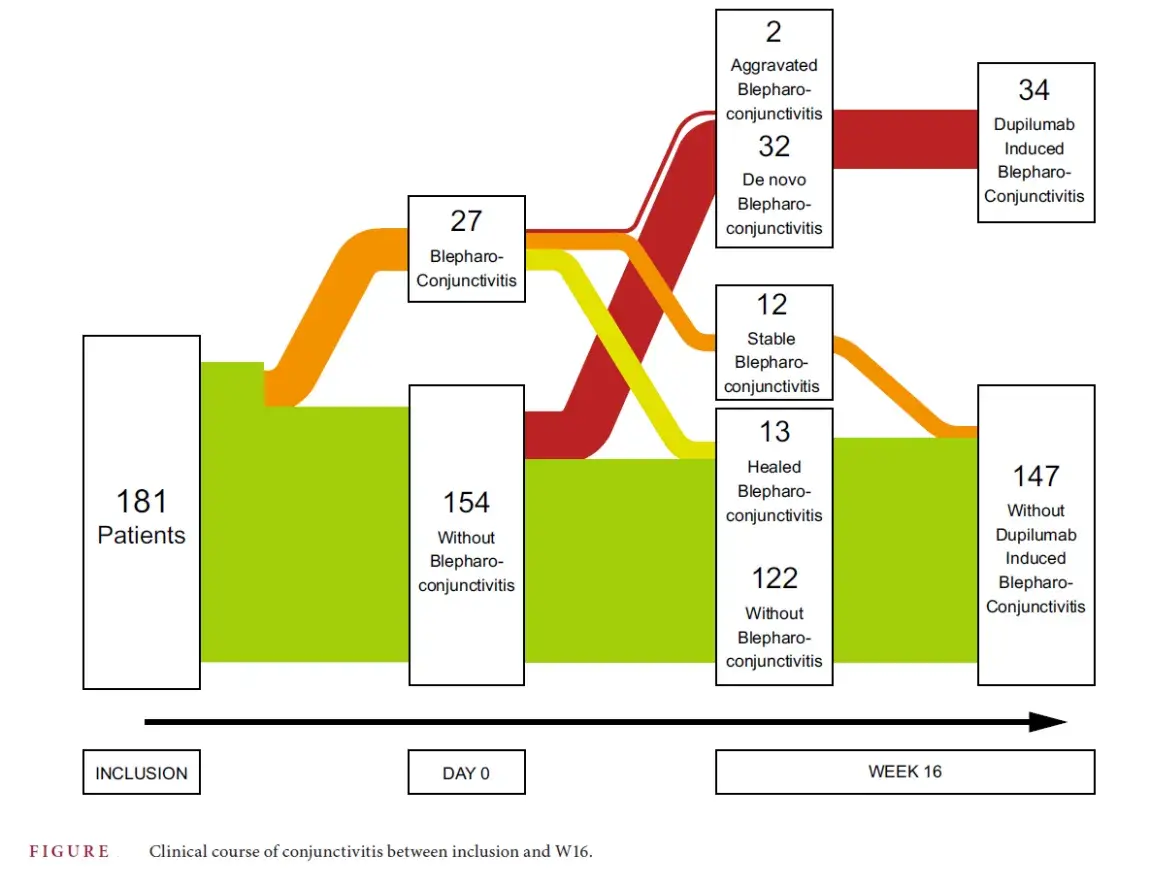
Most events were moderate. A multivariate analysis showed that head and neck AD, erythroderma and the presence of dry eye syndrome at treatment initiation were independent factors associated with dupilumab-induced blepharoconjunctivitis.
To conclude, this study showed that most dupilumab-induced blepharoconjunctivitis cases are of spontaneous onset. AD severity and conjunctivitis at baseline were not found to be associated risk factors in this study.
Based on these initial results with DUPILUMAB, we wanted to apply the same methodology to a new biotherapy recently available for the treatment of AD: TRALOKINUMAB, the study is currently being set up.

